Haema 2019; 10(2):136-139
Konstantinos Liapis
Consultant Haematologist, Georgios Gennimatas Hospital
Full PDF | ![]()
Α. AFRICAN TRYPANOSOMIASIS
1. Cycle of T. brucei(Figure S1)1,2
(A) Τrypomastigote stages (2) of Τ. brucei are produced in the blood and tissue spaces of man when he is bitten by a tsetse fly. The parasites may be found in lymph nodes (3) as well as in the blood. (Β) Τrypomastigotes ingested by the tsetse fly (Glossina spp.) transform into epimastigotes (4) which divide (5) during a complex migration in the fly, eventually forming metacyclic trypomastigotes (6). These can infect another man or reservoir animals (C) in which the blood and tissues are invaded as in man. The disease in domestic cattle is called Nagana.
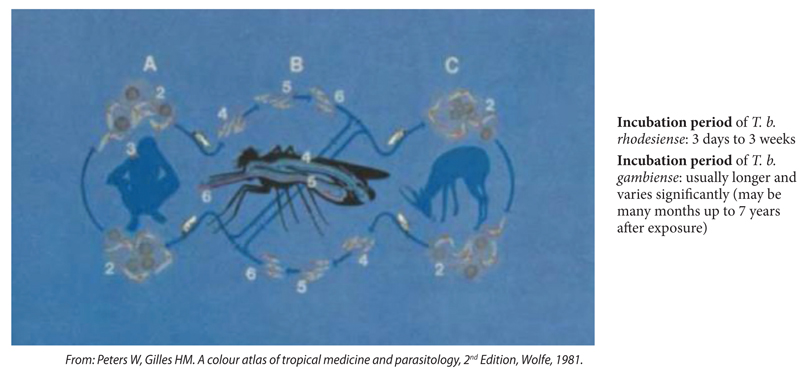
Figure S1. Life cycle of T. brucei.
2. Distribution map of African trypanosomiasis (Figure S2)1-4
Tanzania, Malawi, Zimbabwe, and Zambia (travel-associated African trypanosomiasis in tourists returning from safari in the Zambezi river) are typical countries of origin for T. b. rhodesiense. South Sudan, Congo, and Angola are hot spots for Τ. b. gambiense.
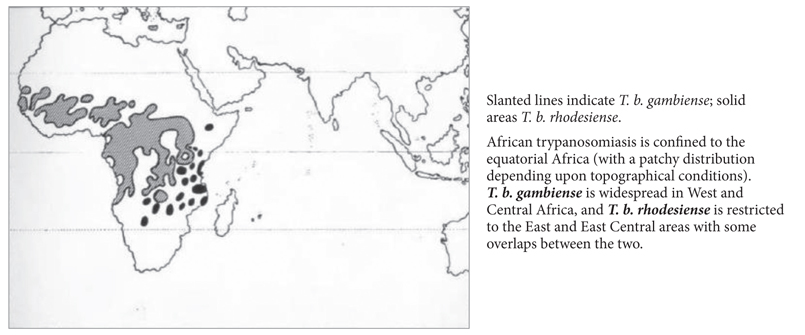
Figure S2. Distribution map of African trypanosomiasis.
3. Clinical information:1-6
The bite reaction, the earliest clinical lesion, is known as the trypanosomal chancre. It resembles a boil (central swelling and erythema over an area up to several cm in diametre) but is usually painless. The diagnosis at this point is made by fluid aspiration from the nodule which contains actively dividing trypanosomes. Trypanosomes appear in the blood one to three weeks after infection. They may be scanty in gambiense but are commonly numerous in rhodesiense infection which is usually a more fulminating disease resembling sepsis.
Disease progresses in 2 stages. In the first stage (haemo-lymphatic phase or lymphoglandular phase), parasites spread in the blood to the lymph nodes, spleen, liver, bone marrow, heart, eyes, and endocrine glands. Enlargement of the lymph nodes, especially in the posterior triangle of the neck (Winterbottom’s sign) is an important clinical feature of T. b. gambiense infection. The clinical picture is frequently that of acute febrile lymphadenitis. Headache, mild splenomegaly and/or hepatomegaly (due to hypertrophy of the reticuloendothelial system), normocytic normochromic anaemia and thrombocytopenia are common. Significant hypergammaglobulinaemia develops in T. b. gambiense due to greatly raised IgM values (this is an important diagnostic finding both for individuals and for field surveys). In some patients, cold agglutinins (IgM antibodies) may be detected in high titre (the blood film shows red-cell autoagglutination). The temperature chart shows irregularly occurring episodes of pyrexia (intermittent fevers) — ddx: malaria, lymphoma, relapsing fever, visceral leishmaniasis (causes fever, generalised lymphadenopathy, and IgG hypergammaglobulinaemia in East and Central East Africa), and brucellosis (also endemic in East and Central East Africa). At this stage the parasites can be detected in thick and thin blood films and lymph nodes aspirates.
In the absence of treatment, the trypanosomes involve the CNS, which leads to the second-stage (meningo-encephalitic phase) of the infection with characteristic lethargy (sleeping sickness). In such instances, the CSF will reveal a lymphocytic pleocytosis, an increased protein content and trypanosomes may be found in stained films of the centrifuge deposit (cytospin). Sometimes morula cells are seen (the morula cells of Mott are IgM producing plasma cells). IgM values are raised in the CSF. At this stage, diagnosis is made by CSF microscopy or serology. In T. b. rhodesiense infection invasion of the CSF may occur within weeks or within four to eight months, whereas several years usually elapse before meningoencephalitis develops in gambiense disease.
Β. AMERICAN TRYPANOSOMIASIS
1. Life cycle of T. cruzi (Figure S3)1,2
Metacyclic trypanosomes (4) of T. cruzi passed in the feaces of infected triatomid bugs (Α) penetrate the skin or mucous membranes e.g. conjunctival mucosa to reach the blood (5) (acute Chagas’ disease). They enter various muscular tissues e.g. cardiac muscle (Β), smooth muscle of the gut (C), or skeletal muscle (D). Here they transfom to amastigotes which divide by binary fission, producing pseudocysts (chronic Chagas’ disease). Subsequently, the daughter amastigotes transform back to trypomastigotes (6, 7). These enter the blood from which they may reinvade muscular tissues, or be picked up by another triatomid bug when it feeds. In the bug the parasites transform to epimastigotes (1-3) which divide in the gut. Finally, some epimastigotes pass to the hindgut where they transform back to infective, metacyclic trypomastigotes (4) which are passed in the feaces as the bug next feeds.

Figure S3. Cycle of T. cruzi in man and triatomid bugs.
2. Distribution of Chagas’ disease (Figure S4)1-4
Chagas’ disease exists in localised endemic zones in Central and South America from the Andes to the Atlantic coast as far south as the latitude of the River Plate. The causative agent T. cruzi may be found in blood films as short C or S shaped trypomastigotes with a prominent kinetoplast, but diagnosis of Chagas’ disease is typically made by serology (a problem is the presence of cross-reactive Abs with L. chagasi). Chagas’ disease is a serious public health problem in South America e.g. 1.5% incidence in Argentina.

Figure S4. Distribution map of Chagas’ disease.
3. Different forms of trypanosomes (Figure S5)7
Trypanosomes occur in several forms, classified on the basis of the position of the kinetoplast into: trypomastigote, epimastigote, and amastigote forms. In the amastigote form, the flagellum is very short and the mitochondrium is poorly developed (Modified from: Mackie TΤ, Hunter GW III, Brooke Worth C. A manual of tropical medicine. WB Saunders Company, Philadelphia, 1945).

Figure S5. Morphology of trypanosomes.
References
- Peters W, Gilles HM. A colour atlas of tropical medicine and parasitology. 2nd ed. London: Wolfe; c1981.
- Peters W, Pasvol G. A colour atlas of tropical medicine and parasitology. 6th ed. London: Mosby Elsevier; c2007.
- Cook GC, Zumla AI. Manson’s tropical diseases. 22nd ed. London: Εlsevier Saunders; c2008.
- Farrar J, Hotez P, Junghanss T, Kang G, Lalloo D, White N. Manson’s tropical diseases. 23rd ed. London: Εlsevier Saunders; c2014.
- Loffler H, Rastetter J, Haferlach T. Atlas of clinical hematology. 6th ed. Berlin Heidelberg: Springer-Verlag; c2005.
- Stich A, Abel PM, Krishna S. Human African trypanosomiasis. BMJ. 2002 Jul;325(7357):203-6.
- Mackie TΤ, Hunter GW III, Brooke Worth C. A manual of tropical medicine. Philadelphia: WB Saunders Company; c1945.