Haema 2019; 10(2):66
Konstantinos Liapis
Consultant Haematologist, Georgios Gennimatas Hospital
Full PDF | ![]()
1. Cycle of W. bancrofti and B. malayi (Figure S1)1,2
Third-stage infective larvae (a) enter the mouthparts of the mosquito from which they penetrate the skin when the insect bites (b). They enter the lymphatics (lymphatic vessels and lymph glands) where the worms mature into thread-like adults (c) which can live for many years and mate. Blockage of lymphatic vessels by the adult worms leads to elephantiasis (d) in some individuals. Larvae (e) are produced in the female worms and transform to microfilariae (f) which enter the peripheral circulation. From there they are picked up by mosquitoes with a blood meal (g). The microfilariae (h) penetrate the insect’s gut wall to develop in its tissues (i, j) in which they mature to the third stage larvae.
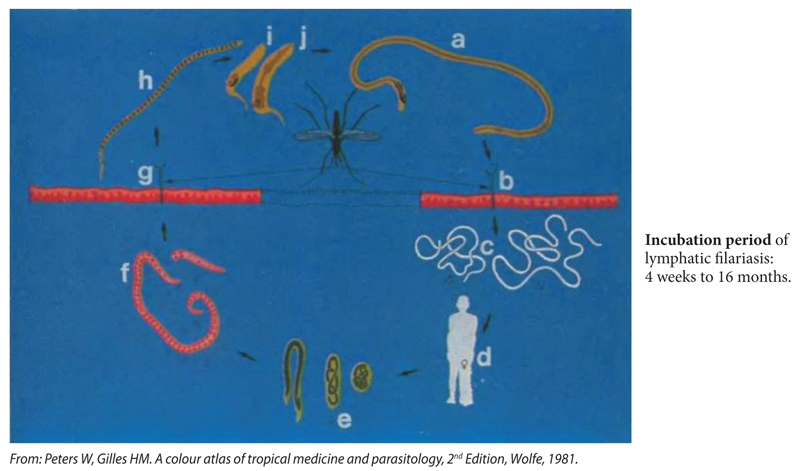
Figure S1. Life cycle of W. bancrofti and B. malayi.
2. Distribution map of W. bancrofti (Figure S2)1-4
W. bancrofti is widely distributed throughout the tropics. A very high humidity level favours transmission of the disease. Although normally nocturnal, a subperiodic diurnal form occurs in the Eastern Pacific e.g. Polynesia, New Caledonia (W. bancrofti var. pacifica). Note: endemic lymphatic filariasis caused by Wuchereria bancrofti is present in Egypt.

Figure S2. Distribution map of W. bancrofti.
Brugia malayi has only been recognised in Asia. The nocturnal periodic from occurs in India, Sri Lanka, South-East Asia, and Japan. The nocturnal subperiodic form occurs only in Malaysia, Borneo, and the Philippines.
3. Cycle of Loa loa (Figure S3)1,2
When the female Chrysops fly bites, the infective third-stage larvae (a) enter the subcutaneous tissue of man (b) where they mature into adults (c) within about one year and they mate. The adults live for 4-12 years. The females (about 7 cm long) migrate through the subcutaneous tissues and may cross the front of the eye under the conjuctiva (d). Microfilariae (f) develop from larvae (e) in the female worn and circulate in the blood with which they are picked up by another fly (g). In the gut of the fly the microfilariae (h) exsheath, penetrate the insect’s gut wall to develop in its tissues first to ‘sausage-like’ forms (i, j), then infective third-stage larvae (a). The larvae infect a new host when the Chrysops takes another blood meal.

Figure S3. Life cycle of Loa loa.
The migration of adult L. loa through subcutaneous and connective tissues of the body may cause allergic symptoms: urticarial rash, pruritus, allergic dermatitis, angioedema, and transient subcutaneous non-pitting oedema usually in the extremities e.g. wrists and forearms known as Calabar swelling (ddx: trauma, arthritis, soft tissue infection, and cellulitis) lasting about three days (up to 15 days). A marked eosinophilia (60-90%) accompanies this phase of the infection.
Loiasis has been associated with renal complications (glomerulonephritis, nephrotic syndrome) and a wide range of neurologic manifestations.
4. Distribution of loiasis (Figure S4)1-4
Loa loa is confined to Africa, extending from the Gulf of Guinea in the West to the Great Lakes in the East. Chrysops fly which transmits loiasis lives in the canopy of primary rainforests.

Figure S4. Distribution map of loiasis.
5. Transmission of filariasis:1-5
– W. bancrofti (in South-East Asia, Ιndia, Central Africa, Egypt, Central and South America, West Indies) and W. bancrofti var. pacifica (in Indonesia) are transmitted by mosquitoes Culex (urban forms), Aedes, and Anopheles.
– B. malayi (in South-East Asia, Ιndia) and B. timori (in South-East Asia) are transmitted by mosquitoes Culex, Aedes, and Anopheles.
Loa loa (only in Africa) is transmitted by Chrysops flies.
– M. perstans (in tropical Africa and South America) is transmitted by Culicoides midges.
– M. ozzardi (in Βrazil, Guiana, Colombia, and the Caribbean) is transmitted by Culicoides and Simulium midges (it is probably non-pathogenic to man).
Note: in 2000, WHO launched the Global Programme to Eliminate Lymphatic Filariasis (GPELF) which has reduced the number of microfilaraemia cases and transmission in some countries (e.g. China, Korea, and Egypt).
6. Clinical information:1-5
Clinical manifestations of lymphatic filariasis (Bancroftian filariasis and Malayan filariasis):
– asymptomatic (subclinical) microfilaraemia (microfilaraemia itself does not produce any symptoms)
– acute adenolymphangitis (ADLA): febrile episodes with chills, headache, lymphangitis and lymphadenitis (the involved limb is warm, oedematous, and the local lymph nodes are enlarged and painful – ddx: erysipelas, cellulitis, DVT, bacterial lymphadenitis, lymphoma). Recurrent episodes of ADLA eventually lead to lymphoedema (i.e. chronic swelling + excess tissue: thickened skin, skin folds, knobs and papillomatosis).
in the acute stages, orchitis may also occur commonly associated with hydrocele.
– complications of chronic lymphatic obstruction include hydrocele(s), lymphocele, lymphadenopathy, lymphoedema (legs, arms, breasts, scrotum), elephantiasis (stage ΙΙΙ chronic lymphoedema), erysipelas or cellulitis, chyluria (the dilated lymph vessels rupture and discharge chyle into the urinay tract), and chylothorax. By the stage of elephantiasis, microfilariae are rarely found in blood films.
– tropical pulmonary eosinophilia (TPE): it presents with dry nocturnal cough, bronchospasm, low-grade fever and hypereosinophilia (>2×109/l). It is more common in those returning from India. The blood film is typically negative for microfilariae serology may also be negative (tropical pulmonary eosinophilia lasts >15 days ≠ Loeffler’s syndrome). The differential diagnosis includes Churg-Strauss syndrome and bronchial asthma (asthma, unlike TPE, is characterised by the recurrent nature of symptoms).
The most common manifestations are hydrocele (usually unilateral), lymphoedema and ADLA. B. malayi is usually milder than W. bancrofti and is not associated with urogenital complications (chyluria). Note: filarial larvae inside lymphatic vessels in various tissues may appear in late, inactive phases as serpiginous calcifications visible on X-rays.
Clinical manifestations of loiasis:
– asymptomatic (subclinical) microfilaraemia (usually in long term residents in endemic regions ≠ visitors).
– allergic reactions (considered to be hypersensitivity reactions to adult worms and dying microfilariae) such as uricaria, angioedema, pruritus (intense pruritus is an early sign), eosinophilia, high serum IgE, Calabar swellings (usually in visitors), and a creeping sensation in the skin.
– L. loa in the eye: the movement of the adult worm under the conjunctiva gives rise to considerable irritation and congestion (ddx: Dirofilaria repens found in Europe and Thelazia callipaeda found in Αsia and Europe)
– complications: glomerulonephritis, nephrotic syndrome, cardiac complications (myocarditis, Davies’ tropical endomyocardial fibrosis, Loeffler’s endocarditis or fibroplastic parietal endocarditis which leads to restrictive cardiomyopathy) caused by eosinophilic heart infiltration, rarely meningoencephalitis, rarely lymphadenopathy and splenomegaly.
Clinical manifestations of Mansonella perstans:
- asymptomatic (subclinical) microfilaraemia
- low-grade fever, abdominal complaints, pruritus, rash, and eosinophilia may occur
- M. perstans infection is usually asymptomatic or mild. Only pruritus and transient rashes, angioedema, or subcutaneous swellings resembling Calabar swellings.
- complications are very rare, usually occurring in HIV (+) patients: meningoencephalitis and eosinophilic meningitis with microfilariae in the CSF.
In tropical Africa Mansonella and Loa loa often co-exist, so you should rule out L. loa in any case positive for Mansonella (L. loa is large, thick, sheathed and has a tapered tail ≠ Mansonella perstans).
References
- Peters W, Gilles HM. A colour atlas of tropical medicine and parasitology. 2nd ed. London: Wolfe; c1981.
- Peters W, Pasvol G. A colour atlas of tropical medicine and parasitology. 6th ed. London: Mosby Elsevier; c2007.
- Cook GC, Zumla AI. Manson’s tropical diseases. 22nd ed. London: Εlsevier Saunders; c2008.
- Farrar J, Hotez P, Junghanss T, Kang G, Lalloo D, White N. Manson’s tropical diseases. 23rd ed. London: Εlsevier Saunders; c2014.
- World Health Organisation. Βench aids for the diagnosis of filarial infections. Geneva: WΗO; c1997.