Haema 2019; 10(2):144-146
Konstantinos Liapis
Consultant Haematologist, Georgios Gennimatas Hospital
Full PDF | ![]()
1. Cycle of leishmaniasis (Figure S1)1,2
Promastigotes (Ι) of various Leishmania species enter the skin of a vertebrate host e.g. dog, man (B) when the female sanfly (A) bites, and transform into amastigotes which are phagocytosed by macrophages (ΙΙ). In the macrophages the amastigotes divide (ΙΙΙ), finally rupturing the host cell. They then enter either macrophages of the reticuloendothelial organs: spleen, liver, bone marrow, lymph nodes (IV) (visceral leishmaniasis), or of the skin (V) (cutaneous leishmaniasis) where they continue to divide by binary fission. Some parasites circulate in monocytes of the blood, and are picked up in these, or with skin macrophages (VI) when another sandfly bites. In the sandfly (A) they transform into promastigotes in the midgut, then migrate within the gut, attach to the gut wall and multiply as promastigotes (VII). Finally they migrate forward to the pharynx and proboscis (VIII) from which they enter the skin of a new vertebrate host when the fly takes another meal.
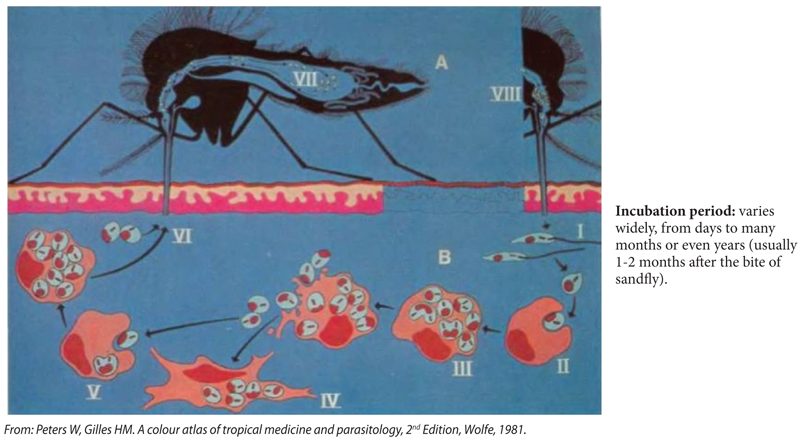
Figure S1. Cycle in man, animals and sandflies.
2. Distribution map of visceral leishmaniasis (FIgure S2)1-5
Visceral leishmaniasis (VL) caused by L. donovanni or L. infantum occurs in the Mediterranean littorals, the Balkans, the Middle East, Sudan, East Africa (Ethiopia, Kenya), the Indian subcontinent (West Bengal, Bangladesh, Nepal, Bhutan), China, Central and South America (‘L. chagasi’). In the Mediterranean, L. infantum is responsible for VL with the exception of Cyprus (L. donovanni). Other species e.g. L. tropica in the Middle East and L. amazonensis in South America are occasionally viscerotropic and may be detected by bone marrow examination.
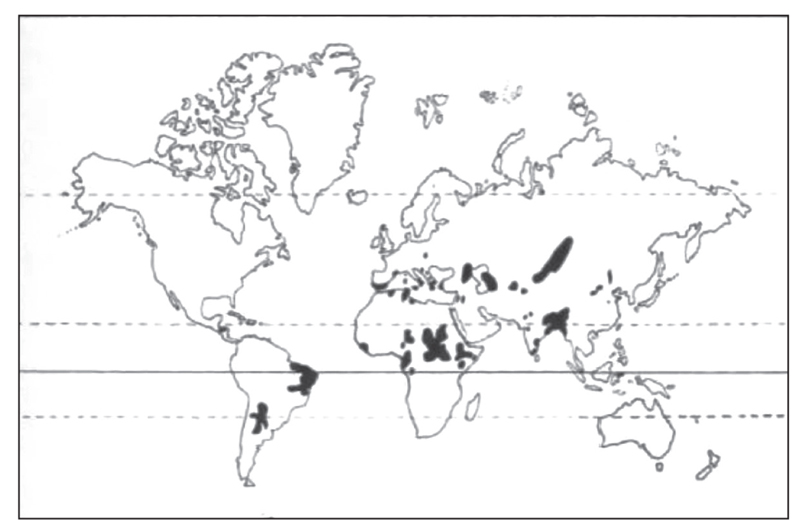
Figure S2. Distribution map of visceral leishmaniasis.
Globally, 60% are children. Children present with irregular fever, anaemia, a moderately enlarged non-tender liver, and a greatly enlarged firm spleen (infantile kala-azar).
Clinical information:1-10
The classic clinical picture is pyrexia, weight loss, splenomegaly, pancytopenia, high ESR, and hypergammaglobulinaemia (Figure S3). However:
– some patients have no fever; others may have low grade fever or intermittent or periodic fever
– the full blood count invariably shows leukopenia or a low-normal leukocyte count (due to neutropenia with or without lymphopenia). Anaemia and/or thrombocytopenia may or may not be present together with leukopenia (in any combination). The degree of anaemia and thrombocytopenia varies from mild to severe.
If the leukocyte count is high or high/normal in a patient with febrile splenomegaly → VL is very unlikely.
– splenomegaly and/or hypergammaglobulinaemia may be absent, especially in the early phase of the disease (acute visceral leishmaniasis) and in immunocompromised hosts.
- VL is followed by hyperplasia of macrophages and lymphocytes, massive production of IgG, and progressive hepatosplenomegaly (kala-azar). The size of the spleen is related directly to the duration of infection and to the severity of pancytopenia.
- Anaemia is usually moderate or severe without any special features. Anaemia is due primarily to hypersplenism (i.e. expansion of plasma volume with haemodilution, splenic sequestration, and haemolysis). Dyserythropoiesis and ineffective erythropoiesis have a role in the causation of anaemia in at least some patients. In India, about half of patients have moderate to severe megaloblastosis due to folate deficiency secondary to increased demands from haemolysis and dietary deficiency.
- Visceral leishmaniasis may rarely be complicated by autoimmune haemolytic anaemia (IgG warm-type, less often cold-type). Coombs’ test is positive in many cases (without evidence of haemolysis). Anti-I cold agglutinins or type III cryoglobulins have been observed.
- Visceral leishmaniasis may mimic haematological malignancy especially lymphoma or atypical myelodysplastic syndrome (cytopenia, dyserythropoiesis). Occasionally, leishmaniasis is diagnosed at splenectomy e.g. splenectomy performed because of suspicion of splenic lymphoma.
- A small band of monoclonal paraprotein or even two M-bands (diclonal gammopathy) may be present (usually ΙgG, less often ΙgΑ) on a background of polyclonal hypergammaglobulinaemia (i.e. no immunoparesis).
- A generalised lymphadenopathy is common in African kala-azar, children, and HIV (+) individuals.
- Dermal lesions which vary in appearance (usually a non-specific papulomacular rash) may be seen in visceral leishmaniasis. In fact, some dermal lesions may contain amastigotes. These lesions should not be confused with cutaneous leishmaniasis which typically manifests as a chronic ulcer with raised borders or with the syndrome of post kala-azar dermal leishmanoid (PΚDL), a sequel to visceral leishmaniasis following treatment in Africa, India, and China which presents with leonine facies (ddx: lepromatous leprosy, cutaneous lymphoma). Dermal lesions in PKDL contain amastigotes in large numbers.
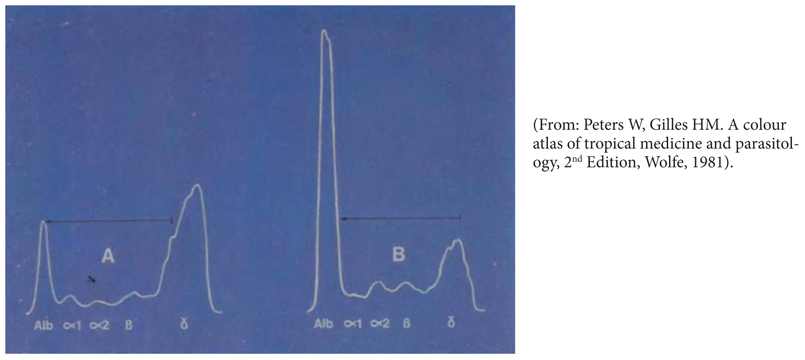
Figure S3. Serum protein electrophoresis before (A) and after treatment (B) of kala-azar. Large quantities of IgG are produced and the albumin:globulin (A/G) ratio is reversed.
Cutaneous leishmaniasis (CL) rarely concerns haematologists. Bone marrow aspiration may be requested in order to exclude visceral disease or when a patient with a history of haematological disease e.g. MDS develops CL. The typical lesion is cutaneous ulcer (s) with raised borders, painless, involving exposed areas (face, especially cheeks and limbs). CL typically begins as a painless papule that enlarges to a nodule with a central crust. The papules and nodules enlarge, develop central ulceration, and take approximately 1 year to heal without treatment. CL is one of the most important considerations in patients with ulcerative lesion(s) returning from the tropics and subtropics. Ηot spots for CL include Afghanistan, Ιran, Pakistan, Persian Gulf, Αlgeria, Tunisia, West Africa, Central American countries (e.g. Belize), and South America (e.g. French Guiana). CL occurs in the Mediterranean littoral (Old world cutaneous leishmaniasis or oriental sore), typically due to L. tropica (minor) which causes ‘dry’ lesion and L. (tropica) major which causes ‘wet’ lesion in rural areas.
References
- Peters W, Gilles HM. A colour atlas of tropical medicine and parasitology. 2nd ed. London: Wolfe; c1981.
- Peters W, Pasvol G. A colour atlas of tropical medicine and parasitology. 6th ed. London: Mosby Elsevier; c2007.
- Cook GC, Zumla AI. Manson’s tropical diseases. 22nd ed. London: Εlsevier Saunders; c2008.
- Farrar J, Hotez P, Junghanss T, Kang G, Lalloo D, White N. Manson’s tropical diseases. 23rd ed. London: Εlsevier Saunders; c2014.
- Christodoulou V, Antoniou M, Ntais P, Messaritakis I, Ivovic V, Dedet JP, et al. Re-emergence of visceral and cutaneous leishmaniasis in the Greek island of Crete. Vector Borne Zoonotic Dis. 2012 Mar;12(3):214-22.
- Loffler H, Rastetter J, Haferlach T. Atlas of clinical hematology. 6th ed. Berlin Heidelberg: Springer-Verlag; c2005.
- Liapis K, Chung Y, Akhtar W, Taussig D. A patient with rheumatoid arthritis, cryoglobulinaemia, and an “accidental” finding. BMJ. 2014 Apr;348:g2701.
- Molina R, Gradoni L, Alvar J. HIV and the transmission of Leishmania. Ann Trop Med Parasitol. 2003 Oct;97 Suppl 1:29-45.
- Desjeux P. The increase in risk factors for leishmaniasis worldwide. Trans R Soc Trop Med Hyg. 2001 May-Jun;95(3):239-43.
- Reithinger R, Dujardin JC, Louzir H, Pirmez C, Bruce Alexander B, Brooker S. Cutaneous leishmaniasis. Lancet Infect Dis. 2007 Sep;7(9):581-96.