Case Report
Haema 2019; 10(1):46-49
Ioanna Sakellari,1 Ioannis Batsis,1 Despina Mallouri,1 Chrysavgi Lalayanni,1 Eleni Gavriilaki,1 Vassiliki Karampekou,2 Panayotis Palladas,2 Ioannis Matzarakis,3 Christos Smias,1 Achilles Anagnostopoulos1
1Haematology Department and BMT Unit, G. Papanicolaou Hospital, 2Department of Radiology, G. Papanicolaou Hospital, 3Laboratory of Pathology, G. Papanicolaou Hospital
Full PDF | ![]()
Abstract
OBJECTIVES: Autologous and allogeneic haematopoietic cell transplantation (HCT) are established therapies for multiple myeloma (MM); however, patients continue to relapse post both types of transplantation. Extra-medullary Plasmacytoma (EM) is defined as proliferation of plasma cells without evidence of MM progression. EM development during MM natural history accounts for less than 5-10%, mainly in the head and neck. EM occurrence in the liver is extremely rare. Clinical presentation of post-transplant relapse is also very heterogeneous and few reports exist. We report one of the few cases of multiple extramedullary relapses and finally fatal hepatic EM plasmacytoma in a young patient who underwent tandem autoHCT and alloHCT for high-risk MM. METHODS: Patient’s clinical record and available literature was reviewed in detail. RESULTS: Our patient succumbed to a very severe, extensive chronic graft-versus-host disease treated mainly with antimyeloma biologic agents and aggressive extramedullary liver plasmacytomas manifesting as end-stage myeloma. CONCLUSIONS: EM relapse post alloHCT is linked to poor prognosis despite aggressive treatment and reflects the underlying disease biology. In the era of novel treatments, the severity of these complications and the diverse therapeutic approaches used in different centers highlight the need of larger, multi-center studies to evaluate current practices.
Key words: Extramedullary plasmacytomas, Haematopoietic transplantation, Multiple myeloma
Corresponding author: Ioanna Sakellari MD, PhD, Haematology Department and BMT Unit, G. Papanicolaou Hospital, Exokhi, 57010 Thessaloniki, Greece, Tel.: +30 2313307550, Fax: +30 2313307521, e-mail: bmt@gpapanikolaou.gr
INTRODUCTION
In the era of novel treatments, multiple myeloma (MM) is considered as a chronic but still incurable disease.1,2 Although autoHCT and alloHCT are established therapies for MM, patients continue to relapse, either as medullary or extra-medullary lesions. Solitary Extramedullary (EM) plasmacytoma is defined as proliferation of plasma cells without MM progression,3-5 Population-based data are scant, more often seen at autopsy. Alexiou et al. published the most extensive series of EM including nearly 900 patients based on reported cases between 1905 and 1995.4 During 1992−2004, 1543 plasmacytoma and 23544 MM cases, a 16-times higher rate, were diagnosed according to SEER (Surveillance, Epidemiology, and End Results) data.5 EM development during MM natural history accounts for less than 5 to 10 % in most series.6-9 Plasmacytoma relapse, occurring either isolated or concurrent with systemic relapse, is rare after alloHCT and autoHCT.10-13
Herein, we report a case of multiple EM relapses and finally fatal liver plasmacytoma in a patient who underwent tandem autoHCT and alloHCT for high-risk MM. Diagnosis and management was complicated by severe chronic graft vs disease (GVHD) and abnormal liver tests at presentation.
CASE PRESENTATION
A young male patient, aged 35, was diagnosed with IgG (λ light chain) MM: stage II -ISS, karyotype 47,XY, +4, +8, dic(1:11)(p11:q25), 13, der(22)t(1:22)(q12:q13)2 /46,XY20 and 13q14 [200 cells] in FISH analysis. Six months later, peripheral autologous graft was collected post 4 cycles of Bortezomib – Vincristine – Liposomal Doxorubicin – Dexamethazone plus Cyclophospamide 4gr/m² mobilization in a very good partial remission (VGPR). He underwent autoHCT and consequently tandem allogeneic sibling transplant post Fludarabin – Melphalan. The patient provided signed informed consent allowing anonymous publication of medical data.
Chimerism studies showed complete donor engraftment by the end of first month. The patient developed subclinical chronic GVHD (positive skin and mouth mucosal biopsies at 3 months) while on cyclosporine administration until 1 year post transplant. Afterwards, he experienced simultaneously extensive chronic GVHD and relapse with monoclonal protein secretion (15%) so corticosteroids, Mycophenolate-Mofetil (MMF) plus Bortezomib were administered for at least 4 months. Then, Bortezomib was stopped after the monoclonal disappearance.
In the meantime, despite adequate standard immunosuppression, moderate to severe pulmonary GVHD was diagnosed. Twenty two months post allotransplant, he experienced the first extramedullary relapse in the skull, which was irradiated and Lenalidomide replaced the MMF administration. Due to severe allergic reaction to Lenalidomide, Bortezomib was re-initiated with continuous use of corticosteroids for 3 months. In the meantime, lung diffusion tests deteriorated, along with pulmonary infections and he was treated with Thalidomide plus Dexamethasone 40mg/week. The PET-CT scan, bone marrow aspiration and biopsy were negative in the 30th month. Due to Thalidomide adverse reactions, in the 38th month post-transplant, the patient started a new treatment which combined the anti GVHD and anti-myeloma strategy, including Lenalidomide – Cyclophosphamide and corticosteroids until the 43th month. Then, he presented with 2 extramedullary sites in the skull which were successfully irradiated.
Three months after a successfully managed severe pulmonary opportunistic infection, the patient presented with profound asthenia and several space-occupying hepatic lesions in the liver. The rest of the examination was unremarkable. However, abdominal ultrasound revealed masses (41.7×37.6×32.2 and 13 mm) in the liver parenchyma, which were also evident in the abdomen MRI scan with similar sizes. CT-guided percutaneous biopsy was found consistent with plasmacytoma (lambda light chain restricted, similar to previously existing monoclonal immunoglobulin pattern at the diagnosis of MM). The liver biopsy immunochemistry was positive for CD138, MUM1, lambda proteins. Liver biopsy revealed a diffuse portal and sinusoidal infiltration of plasma cells with lambda light-chain restriction (Figure 1). Bone marrow infiltration, serum protein electrophoresis and immunofixation electrophoresis were negative.
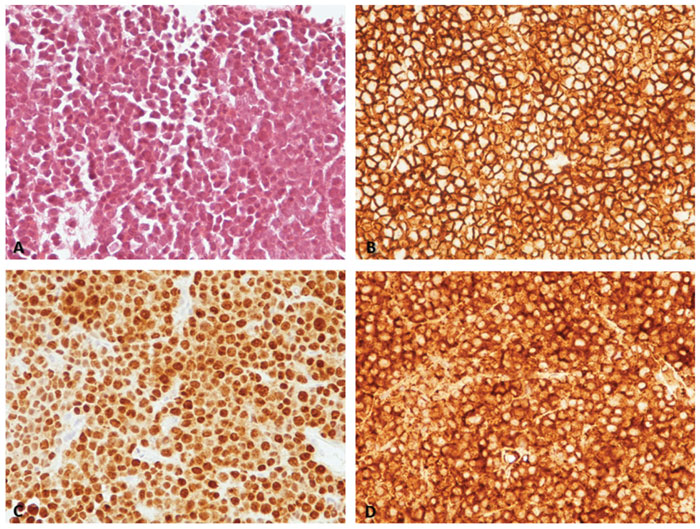
Figure 1. A. Histopathology examination of the biopsy material revealed total infiltration of half of the cylinders by a neoplasm, consisting of pleomorphic forms of plasma cells, mainly immature and anaplastic, whereas the mature plasma cells are rare. The neoplastic cells are arranged in loose groups with no specific pattern. (H&E x400). B. C. and D. Immunohistochemistry showed positivity of these cells for CD138 (B) and MUM1 (C). A lambda (λ) light-chain monoclonality was revealed (D), with only scarce expression of immunoglobulin light κ-chains, immature and anaplastic, whereas the mature plasma cells are rare.
Initial laboratory values included elevated liver function tests (aspartate aminotransferase 157 U/L and alanine aminotransferase 351 U/L, bilirubin 2.1 mg/dl, direct bilirubin 1.8 mg/dl, gamma-glutamyl transferase 4109 U/L) without systemic or renal disease attributable to myeloma. Bearing in mind the patient’s comorbidity, he received one course of Melphalan (80 mg intravenously), Pomalidomide and corticosteroids without DLIs due to GVHD. The patient suffered from recurrent severe infections, multi-organ failures and chemotherapy or novel agent was ceased. Eventually, the patient succumbed six months after the diagnosis of EM liver plasmacytomas.
DISCUSSION
In summary, the present case showed a puzzling relapse pattern involving hepatic plasmocytomas in a patient with multiple, mainly extramedullary, relapses despite extensive GVHD (pulmonary, eyes, skin sclerotic lesions).
To the best of our knowledge, there are less than 10 reported cases of liver plasmacytoma and diffuse hepatic infiltration.14-21 Among rare plasma cell dyscrasias, the present case is one of the few cases of MM presenting as acute liver disease. Besides its complexity, diagnosis requires biopsy confirmation and extensive testing, that will allow differential diagnosis from reactive plasmacytoma, plasma cell granuloma and MALT, marginal zone, and immunoblastic lymphomas.9
In the report of the Greek myeloma study group in 97 patients with EM plasmacytoma, Katodritou and colleagues8 concluded that immunoparesis at diagnosis was the most powerful factor of progression to MM. This is in line with our patient’s status of deep immunosuppression, where profound immunoparesis accompanied solitary plasmacytomas despite immunoglobulin infusions.
Beyond profound immunoparesis, concerns have been raised that adoption of thalidomide, lenalidomide, bortezomib and increased numbers of HCT might contribute to increased EM rates.22 There is one report of liver plasmacytoma among others which showed EM relapse of MM after thalidomide.23 In addition, Cerny et al. reported six fatal cases of extramedullary relapses of multiple myeloma (MM) pretreated with immunomodulatory therapy and/or HCT.24 On the contrary, Vagra et al. concluded that there is no evidence that bortezomib-lenalidomide-based front-line therapy precipitates EM.25
In conclusion, our case illustrates the clinical aggressiveness of relapsed MM post transplantation leading to poor outcomes despite aggressive treatment approaches. Increased clinical awareness and novel individualized approaches are needed to deal with the problem of extramedullary disease recurrence with an ominous outcome. The severity of these complications and the diverse therapeutic approaches used in different centers highlight the need of larger, multi-center studies to evaluate current practices.
Conflict of Interest and Sources of Funding
None.
REFERENCES
1. Singhal S, Mehta J. Novel therapies in multiple myeloma. International journal of hematology. 2003 Apr;77(3):226-31.
2. Laubach J, Garderet L, Mahindra A, Gahrton G, Caers J, Sezer O, et al. Management of relapsed multiple myeloma: Recommendations of the International Myeloma Working Group. Leukemia. 2016 May;30(5):1005-17.
3. Dimopoulos MA, Moulopoulos LA, Maniatis A, Alexanian R. Solitary plasmacytoma of bone and asymptomatic multiple myeloma. Blood. 2000 Sep;96(6):2037-44.
4. Alexiou C, Kau RJ, Dietzfelbinger H, Kremer M, Spiess JC, Schratzenstaller B, et al. Extramedullary plasmacytoma: tumor occurrence and therapeutic concepts. Cancer. 1999 Jun; 85(11):2305-14.
5. Dores GM, Landgren O, McGlynn KA, Curtis RE, Linet MS, Devesa SS. Plasmacytoma of bone, extramedullary plasmacytoma, and multiple myeloma: incidence and survival in the United States, 1992-2004. Br J Haematol. 2009 Jan;144(1):86-94.
6. Damaj G, Mohty M, Vey N, Dincan E, Bouabdallah R, Faucher C, et al. Features of extramedullary and extraosseous multiple myeloma: a report of 19 patients from a single center. Eur J Haematol. 2004 Dec;73(6):402-6.
7. Papanikolaou X, Repousis P, Tzenou T, Maltezas D, Kotsopoulou M, Megalakaki K, et al. Incidence, clinical features, laboratory findings and outcome of patients with multiple myeloma presenting with extramedullary relapse. Leuk Lymphoma. 2013 Jul;54(7):1459-64.
8. Katodritou E, Terpos E, Symeonidis AS, Pouli A, Kelaidi C, Kyrtsonis MC, et al. Clinical features, outcome, and prognostic factors for survival and evolution to multiple myeloma of solitary plasmacytomas: a report of the Greek myeloma study group in 97 patients. Am J Hematol. 2014 Aug;89(8):803-8.
9. Weber DM. Solitary bone and extramedullary plasmacytoma. Hematology Am Soc Hematol Educ Program. 2005;373-6.
10. Chong G, Byrnes G, Szer J, Grigg A. Extramedullary relapse after allogeneic bone marrow transplantation for haematological malignancy. Bone Marrow Transplant. 2000 Nov; 26(9):1011-5.
11. Byrne JL, Fairbairn J, Davy B, Carter IG, Bessell EM, Russell NH. Allogeneic transplantation for multiple myeloma: late relapse may occur as localised lytic lesion/plasmacytoma despite ongoing molecular remission. Bone Marrow Transplant. 2003 Feb;31(3):157-61.
12. Alegre A, Granda A, Martinez-Chamorro C, Diaz-Mediavilla J, Martinez R, Garcia-Larana J, et al. Different patterns of relapse after autologous peripheral blood stem cell transplantation in multiple myeloma: clinical results of 280 cases from the Spanish Registry. Haematologica. 2002 Jun;87(6):609-14.
13. Terpos E, Rezvani K, Basu S, Milne AE, Rose PE, Scott GL, et al. Plasmacytoma relapses in the absence of systemic progression post-high-dose therapy for multiple myeloma. Eur J Haematol. 2005 Nov;75(5):376-83.
14. Ghobrial PM, Goel A. Plasmacytoma of Liver Mimicking Hepatocellular Carcinoma at Multiphasic Computed Tomography Evaluation. J Comput Assist Tomogr. 2015 Jul-Aug;39(4):510-2.
15. Lopes da Silva R, Monteiro A, Veiga J. Non-secretory multiple myeloma relapsing as extramedullary liver plasmacytomas. J Gastrointestin Liver Dis. 2011 Mar;20(1):81-3.
16. Thiruvengadam R, Penetrante RB, Goolsby HJ, Silk YN, Bernstein ZP. Multiple myeloma presenting as space-occupying lesions of the liver. Cancer. 1990 Jun;65(12):2784-6.
17. Yagci M, Sucak GT, Akyol G, Haznedar R. Hepatic failure due to CD3+ plasma cell infiltration of the liver in multiple myeloma. Acta haematologica. 2002 Jan;107(1):38-42.
18. Bhandari MS, Mazumder A, Vesole DH. Liver involvement in multiple myeloma. Clinical lymphoma & myeloma. 2007 Sep;7(8):538-40.
19. Petrucci MT, Tirindelli MC, De Muro M, Martini V, Levi A, Mandelli F. Extramedullary liver plasmacytoma a rare presentation. Leukemia & lymphoma. 2003;44(6):1075-6.
20. Husarić S, Pašić J, Alić E, Kuljanin M. Solitary extramedullary plasmocytoma of the liver. Acta medica academica. 2013; 42(1):85-6.
21. El Maaroufi H, Doghmi K, Rharrassi I, Mikdame M. Extramedullary plasmacytoma of the liver. Hematology/oncology and stem cell therapy. 2012 Jul;5(3):172-3.
22. Wirk B, Wingard JR, Moreb JS. Extramedullary disease in plasma cell myeloma: the iceberg phenomenon. Bone Marrow Transplant. 2013 Jan;48(1):10-8.
23. Singhal S, Mehta J, Desikan R, Ayers D, Roberson P, Eddlemon P, et al. Antitumor activity of thalidomide in refractory multiple myeloma. N Engl J Med. 1999 Nov;341(21):1565-71.
24. Cerny J, Fadare O, Hutchinson L, Wang SA. Clinicopathological features of extramedullary recurrence/relapse of multiple myeloma. Eur J Haematol. 2008 Jul;81(1):65-9.
25. Varga C, Xie W, Laubach J, Ghobrial IM, O’Donnell EK, Weinstock M, et al. Development of extramedullary myeloma in the era of novel agents: no evidence of increased risk with lenalidomide-bortezomib combinations. Br J Haematol. 2015 Jun;169(6):843-50.
Received 27 Nov 2017
Accepted 15 Dec 2017